Astounding New Developments in Endoscopic Minimally Invasive Spine Surgery
Joseph Spine Institute, spearheaded by Dr. Samuel A. Joseph, Jr. Is excited to announce a groundbreaking collaboration with Spineology, a leading innovator in spine surgery technologies.
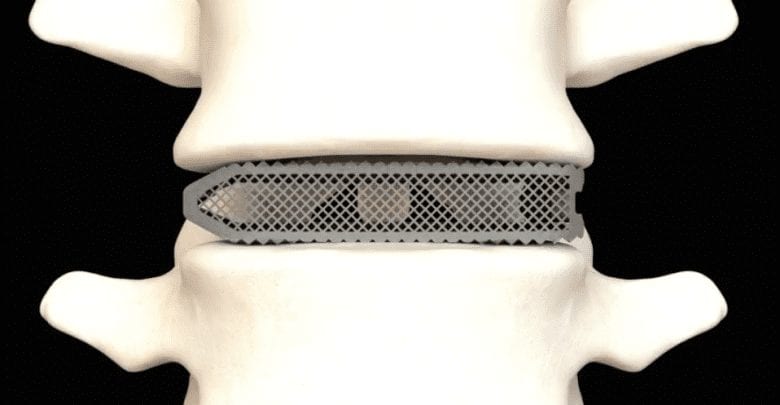
Spineology’s Pioneer Project has brought forth a paradigm shift in the world of endoscopic minimally invasive spine surgery, taking it to an entirely new level.
About the Pioneer Project:
The Pioneer project is the result of a cutting-edge partnership that has redefined the boundaries of endoscopic spine surgery. Spineology and Joseph Spine Institute, spearheaded by Dr. Samuel A. Joseph, Jr. have united to develop innovative products and technologies, achieving a level of precision, minimally invasiveness, and safety previously unattainable in this field.
Key Achievements: Through the relentless dedication and shared vision of our teams, the Pioneer project has introduced ultra minimally invasive endoscopic spine surgery to the forefront of medical science. This transformative achievement promises improved patient experiences, shorter recovery times, and reduced post-operative discomfort, setting a new standard for spine surgery globally.
The Power of Collaboration: Spineology’s reputation for excellence and unwavering commitment to innovation, combined with the visionary leadership of Dr. Samuel A. Joseph and the Joseph Spine Institute, has culminated in a revolutionary leap forward in the realm of spine surgery.
Expressing Our Gratitude: We extend our profound appreciation to the teams at Spineology, Joseph Spine Institute, and Dr. Samuel A. Joseph for their unwavering dedication and outstanding contributions. This partnership has not only advanced the frontiers of medical science but has also made a profound impact on patients seeking minimally invasive spine surgery.
A Legacy of Innovation: Spineology is consistently at the forefront of technological advancements in spine surgery. Testing and re-testing. Imagining and re-imagining. Continually enhancing what spine surgeons will be capable of tomorrow, by revolutionizing the tools they hold in their hands, today. Because when patients trust you with their spine, trusting in your hands is essential. And trusting what’s in your hands is absolutely vital. You can’t wait for the future to provide the latest and greatest. That’s why we have and always will bring you tomorrow’s tools, today.
The Future of Spine Surgery: The Pioneer project represents our shared commitment to reshaping the landscape of spine surgery. This achievement is not merely about cutting-edge technology; it is about transforming the lives of patients and setting a new gold standard in medical care. We invite you to celebrate this remarkable achievement and stay updated with our ongoing journey to pioneer the future of minimally invasive spine surgery.